OECD TG 421: Reproduction/Developmental Toxicity Screening Test
In the early stages of product development or during initial regulatory assessments, identifying reproductive and developmental risks is a top priority. Adhering to OECD TG 421 guidelines and GLP principles, we offer efficient screening services to help you understand the potential impacts of a test substance on fertility and offspring development before committing to more complex, long-term studies.
Study Objective
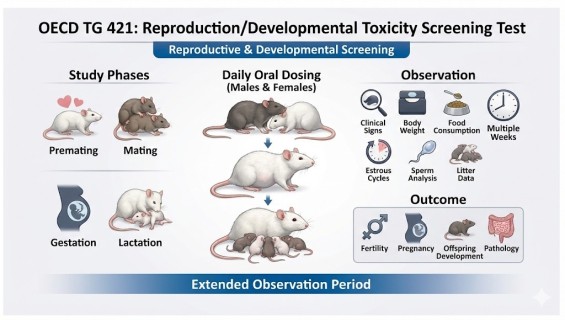
To provide preliminary information on the effects of a test substance on male and female reproductive performance (e.g., gonadal function, mating behavior, conception) and the early development of offspring (e.g., viability, body weight, anatomical abnormalities). This screening is also used to guide dose selection for future long-term studies, such as TG 443.
Service Highlights & Technical Specifications
-
Efficient Screening: Offers a shorter study duration compared to full multi-generation studies, providing key safety indicators in a more cost-effective manner.
-
Reproductive Cycle Coverage: Dosing and observation cover pre-mating, mating, gestation, and the first 13 days of pup life.
-
Endocrine Indicator Tracking: Monitoring maternal estrous cycles, offspring sex ratios, and parental organ weights to capture signs of potential endocrine disruption.
-
Initial Developmental Assessment: Evaluating the survival, clinical signs, and gross necropsy findings of the F1 generation.
Study Procedure
-
Parental Dosing: Males are treated for a minimum of 4 weeks (to cover the full spermatogenic cycle); females are treated starting 2 weeks prior to mating.
-
Mating & Gestation: 1:1 pairing with continuous maternal exposure throughout pregnancy and lactation.
-
Offspring Observation: Recording the number of live births, sex ratio, and pup weights on post-natal days 4 and 13.
-
Necropsy & Pathology: Gross necropsy and preliminary histopathological screening of parental reproductive organs.
Targeted Industries
-
Regulatory Chemical Notification (REACH Annex VIII)
-
Early-stage Risk Screening in Product Development
-
Initial Toxicity Characterization for Pesticides or Industrial Raw Materials
-
Optimization of Subsequent High-level Toxicological Study Designs
Click to Pay
