OECD TG 475 — Mammalian Bone Marrow Chromosomal Aberration Test (In Vivo)
In the biological safety evaluation of medical devices and chemical substances, preventing genetic damage or carcinogenic risks is a core regulatory pillar. OECD TG 475 (Mammalian Bone Marrow Chromosomal Aberration Test) is a rigorous In Vivo cytogenetic assay designed to identify whether a test substance induces structural chromosomal aberrations in the bone marrow cells of laboratory animals. It is recognized by global regulatory bodies as a critical indicator for genotoxicity assessment.
1. Why is OECD TG 475 Crucial for Your Product?
According to international ISO 10993-3 (Genotoxicity, Carcinogenicity, and Reproductive Toxicity) standards and the safety framework of the Taiwan Medical Device Act, medical devices with high exposure risks (e.g., long-term implants or systemic circulation contact) must undergo a comprehensive Genotoxicity Testing Battery.
-
Detection of Clastogenicity: This study directly observes whether a test substance causes chromosome breaks, translocations, or exchanges within a living system. Compared to In Vitro assays, it better reflects the true toxicity of a substance after metabolism and systemic distribution.
-
Assessment of Reproductive and Carcinogenic Potential: Bone marrow is one of the most proliferative tissues in the body. Chromosomal aberrations in these cells are highly correlated with early oncogenesis and potential reproductive system damage.
-
Supporting High-End Market Access (STED): For Class II and III medical devices seeking Quality System Documentation (QSD) approval in Taiwan or submission to the US FDA and EU MDR, In Vivo chromosomal aberration data provides robust evidence of the "genetic safety" of the materials.
2. Technical Depth: Precision Design of OECD TG 475
The execution of OECD TG 475 demands exceptional micro-manipulation and cytogenetic interpretation expertise:
-
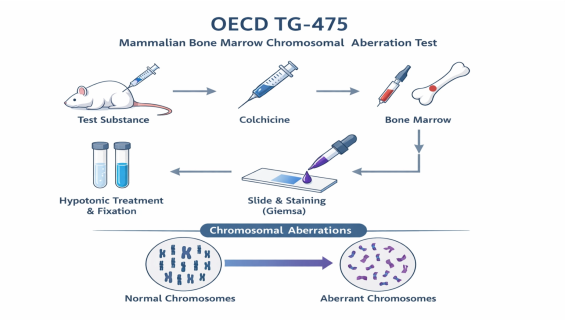
In Vivo Dosing and Sampling Timeline: Typically utilizing rodents, the test substance is administered via the clinical route. The timing of tissue harvesting is critical, as it must align with the most sensitive stage of cell division to capture potential damage.
-
Metaphase Arrest: A mitotic inhibitor (such as Colchicine) is administered prior to sampling to arrest cells in metaphase, allowing for clear visualization of chromosome morphology.
-
Advanced Staining and Preparation: Through specialized spreading, fixation, and Giemsa staining, bone marrow cells are prepared for high-magnification microscopic analysis.
Expert Morphological Analysis: The Weshin expert team rigorously scans at least 200 metaphases per animal to precisely distinguish between aberrations such as gaps, breaks, exchanges, and rings, while calculating the Mitotic Index to evaluate cytotoxicity.
3. Your Premier Regulatory and Testing Partner
Executing OECD TG-475 requires top-tier cytogenetic expertise and professional pathological analysis. We provide:
-
GLP-Compliant Excellence: Our protocols strictly adhere to OECD and ICH guidelines. The resulting data carries international mutual recognition (MAD), serving as a powerful asset for submissions to agencies like FDA and ECHA.
-
High-Sensitivity Analytical Platforms: Combining advanced slide preparation with professional manual microscopic review, we ensure that even minor chromosomal abnormalities are identified.
-
Comprehensive Toxicological Consulting: Our team of experts assists in interpreting complex aberration frequencies and provides strategic regulatory guidance based on study outcomes to ensure your product’s successful market access.
Click to Pay
