OECD TG-488 — Transgenic Rodent Somatic and Germ Cell Gene Mutation Assays
At the highest levels of safety evaluation for medical devices and chemicals, determining whether a substance induces permanent genetic sequence alterations in vivo is the key to clearing rigorous regulatory hurdles. OECD TG 488 represents the pinnacle of modern toxicology for in vivo gene mutation detection, bridging the gap left by traditional assays regarding organ specificity and germ cell toxicity.
1. Why High-Risk Products Require OECD TG 488 Studies?
While not a routine requirement, OECD TG 488 is a critical asset for international market authorizations (such as US FDA PMA/510k, EU MDR/REACH) in the following scenarios:
-
In-depth Validation for Target Organs: Traditional in vivo assays (e.g., Micronucleus Test) primarily observe bone marrow. OECD TG 488 allows for the extraction of any tissue (liver, kidney, lung, brain, etc.) to detect mutations, providing irreplaceable diagnostic value for substances with specific target-organ toxicity.
-
The Last Line of Defense for Heritable Damage: This assay can simultaneously detect mutations in germ cells (sperm or oocytes). If your product involves long-term implantation or could affect reproductive health via systemic circulation, this is the strongest scientific evidence to prove no "heritable damage" occurs.
-
Clarifying In Vitro Positive Results: When a foundational Ames Test yields a positive result and other routine in vivo tests cannot adequately explain the risk, the point mutation data provided by TG 488 can definitively determine if the substance is truly mutagenic in vivo.
2. Technical Depth: Principles of Transgenic Technology
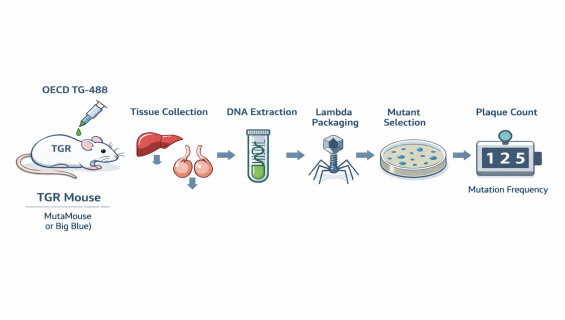
The uniqueness of OECD TG 488 lies in the use of specialized "Transgenic Rodents" (e.g., Muta™Mouse or Big Blue®). The methodology is as follows:
-
Integration of Reporter Genes: Every cell in the test animal carries neutral exogenous reporter genes (e.g., lacZ or cII). These genes do not affect the animal's physiology but precisely record mutations caused by the test substance.
-
In Vitro Recovery and Screening: After the animal is exposed (typically for 28 days), target tissues are harvested. DNA is extracted, and the reporter genes are "recovered" via packaging extracts and transferred into E. coli.
-
Calculation of Mutation Frequency (MF): Through selective screening on specific media, the proportion of mutated genes is calculated. This integration of "in vivo exposure" and "molecular cloning" provides high-sensitivity genotoxicity data.
3. Your Premier Regulatory and Testing Partner
Executing OECD TG 488 requires significant technical expertise and specialized transgenic animal resources. We provide the following values:
-
GLP-Compliant Data Excellence: Our reports strictly adhere to international GLP standards. With international mutual recognition, our data is your best solution for responding to technical inquiries (AI) from global regulatory bodies.
-
Customized Study Design: We recommend the most appropriate "tissue sampling combinations" and "expression windows" tailored to your product’s route of administration to obtain the most representative data.
-
Regulatory Consulting for Market Access: We assist in transforming complex molecular data into professional narratives that persuade regulators, ensuring your product’s successful global compliance and market entry.
Click to Pay
