OECD TG-443 — Extended One-Generation Reproductive Toxicity Study (EOGRTS)
In the elite echelons of chemical safety and regulatory oversight, accurately assessing the "transgenerational" health impact of a substance is pivotal for clearing the world’s most stringent reviews, such as EU REACH high-tonnage registrations. OECD TG-443 (EOGRTS) represents the pinnacle of modern toxicology in reproductive and developmental safety assessment, delivering exhaustive scientific data on reproductive, neurological, and immunological development while strictly adhering to the 3Rs (Reduction, Refinement, Replacement) principle.
1. Why High-Compliance Products Require OECD TG-443 Studies?
This is not a routine test, but a "one-stop" high-level assessment designed for high-volume or biologically active substances. OECD TG-443 is an indispensable asset in the following scenarios:
-
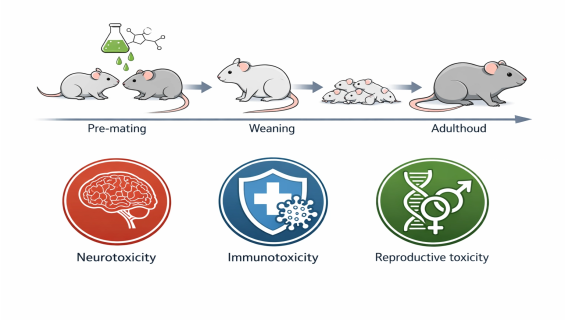
Integrated Deep Validation Across Systems: Unlike traditional studies, EOGRTS allows for the simultaneous detection of Developmental Neurotoxicity (DNT) and Developmental Immunotoxicity (DIT) within a single study framework, determining if a substance impairs brain development or immune defense mechanisms in offspring.
-
Flexible Cohort-Based Design: Depending on initial findings, the study can dynamically trigger the extension of the F1 generation to F2. This "triggered" approach provides irrefutable scientific evidence regarding Endocrine Disrupting (ED) properties.
-
Meeting High-Tonnage Regulatory Demands: It is a core requirement under EU REACH for substances produced at 1000 tonnes or more per year and is a decisive factor in determining if a substance is categorized as a Substance of Very High Concern (SVHC).
2. Technical Depth: Full-Cycle Exposure and Multi-Dimensional Monitoring
The uniqueness of OECD TG-443 lies in its rigorous experimental design and comprehensive coverage of the life cycle:
-
Continuous Exposure Pathway: The parental (P) generation is dosed prior to mating through gestation and lactation; the offspring (F1) are exposed in utero via the mother and directly after weaning, ensuring no critical developmental window is overlooked.
-
Multi-Tiered Cohort Assessment:
-
Cohort 1: Evaluates basic reproductive and developmental parameters (e.g., organ weights, histopathology, sexual maturation).
-
Cohort 2 (DNT): Focuses on the nervous system, including brain morphometry and behavioral functional assessments.
-
Cohort 3 (DIT): Evaluates the primary immune response to antigens in the offspring.
-
-
Endocrine-Sensitive Endpoints: By monitoring Anogenital Distance (AGD), nipple retention, and thyroid hormone levels, the study precisely screens for hormone-like or hormone-disrupting activities.
Click to Pay
