OECD TG-453 — Combined Chronic Toxicity/Carcinogenicity Studies
When facing the world’s most stringent chemical and pharmaceutical regulatory reviews, efficiently and
comprehensively defining a substance's long-term safety is the core challenge for R&D and compliance departments. OECD TG 453 is a powerful integrated study that identifies both long-term organ damage (chronic toxicity) and tumor-inducing risks (carcinogenicity) in a single experimental framework, providing the most complete life-cycle safety endorsement for your product.
1. Why High-Level Compliance Requires OECD TG-453 Studies?
This is not a routine test; it is the "Ultimate Safety Assessment" for substances with widespread exposure risks:
-
Maximizing Data Output: Within a single study design, it generates dual data sets—one for deriving the NOAEL and another for assessing carcinogenic potential—minimizing interpretive gaps caused by biological variation between separate studies.
-
Optimal Balance of Resources and Animal Welfare: By integrating cohorts, TG-453 utilizes animal resources more efficiently than conducting two separate studies, aligning with international 3Rs (Reduction, Refinement, Replacement) principles.
-
Meeting High-Tonnage Regulatory Demands: For high-production-volume chemicals (e.g., REACH >1000 tonnes), active pesticide ingredients, or chronic pharmaceuticals, this study is the decisive basis for regulatory market access.
2. Technical Depth: Rigorous Monitoring of Dual Objectives
The scientific precision of OECD TG 453 lies in its design for different observational endpoints:
-
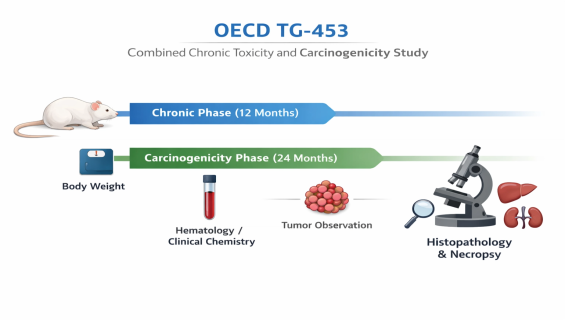
Satellite Groups & Tiered Observation: The study typically includes a 12-month satellite group for chronic toxicity assessment (including biochemical and hematological analysis) and a main group for carcinogenicity observation lasting up to 24 months.
-
Comprehensive Toxicological Scanning: Clinical signs, ophthalmology, urinalysis, and clinical chemistry are monitored throughout the study. Upon termination, detailed histopathological examinations of over 40 organs are performed.
-
Correlation of Dose-Response and Tumor Evolution: Simultaneously analyzes non-neoplastic lesions (e.g., chronic inflammation, degenerative changes) alongside the development of neoplasms (benign and malignant) to clarify the Mode of Action (MoA).
3. Your Premier Regulatory and Testing Partner
Executing OECD TG 453 requires exceptional laboratory management stability and the precise diagnosis of veteran pathologists. We provide:
-
GLP-Compliant Integrated Reporting: Throughout the 24-month study cycle, we ensure total data integrity and consistency. Our integrated reports possess international mutual recognition (MAD), accepted by ECHA, FDA, and EPA.
-
Expert Carcinogenic Risk Interpretation: We help clarify whether observed tumors result from a non-genotoxic mechanism with a defined threshold or a direct genotoxic mechanism—critical for subsequent human health and environmental risk assessments.
-
One-Stop Regulatory Consulting: From range-finding studies to final dossier preparation, we assist in transforming complex data into authoritative regulatory narratives, significantly accelerating your path to market.
Click to Pay
