OECD TG-215 — Fish, Juvenile Growth Test
In environmental toxicology, whether a substance "quietly" inhibits the development rate of an organism is often more consequential than immediate lethality. OECD TG 215 is specifically designed to assess the effects of chemicals on fish during their rapid growth phase. By precisely quantifying changes in weight gain, it detects interferences in metabolism and energy allocation, serving as a core pillar for sub-chronic environmental risk assessment.
1. Why Does Your Product Require an OECD TG-215 Study?
When preliminary screenings indicate potential bioaccumulation or persistence, OECD TG 215 becomes a critical requirement for a robust Environmental Risk Assessment (ERA):
-
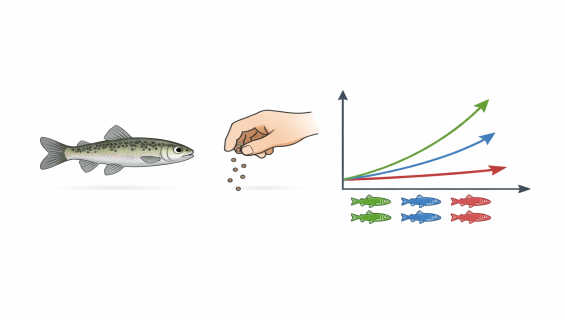
Capturing Sub-lethal Effects: Many substances do not cause mortality at low concentrations but significantly stunt growth. TG 215 focuses on "growth" as a sensitive endpoint, providing finer toxicity thresholds than acute tests.
-
Calculating Specific Growth Rate (SGR): Utilizing precise mathematical models to determine the SGR, this test providing regulatory bodies with a quantified dose-response curve to evaluate impacts on long-term population competitiveness.
-
Bridging Regulatory Data Gaps: For chemicals that do not require full life-cycle testing but need more depth than a 96-hour acute test, TG 215 offers a high-efficiency solution balancing cost-effectiveness with data integrity.
2.Technical Depth: Monitoring Growth Rates and Metabolic Interference
The scientific core of OECD TG 215 lies in tracking the physiological growth trajectory of juvenile fish over a 28-day exposure period:
-
Selecting Critical Growth Phases: The test utilizes juvenile fish (such as Rainbow Trout or Zebrafish) during their linear growth stage, a period highly sensitive to nutritional uptake and chemical interference.
-
Strict Control of Rations and Environment: To ensure growth variance is caused by the "test substance" and not environmental factors, we strictly standardize feeding (fixed percentage of body weight) and water quality parameters.
-
Kinetic Data Analysis: Upon study completion, the NOEC (No Observed Effect Concentration) and LOEC are calculated by comparing weight increments against the control group. This data reveals the potential for metabolic or endocrine disruption.
Click to Pay
