OECD TG 402: Acute Dermal Toxicity Test
When evaluating health risks associated with skin contact, acute dermal toxicity data is a core requirement for regulatory classification and labeling. We provide professional testing services in strict accordance with OECD TG 402 guidelines and GLP principles, ensuring your product’s compliance in international markets.
Study Objective
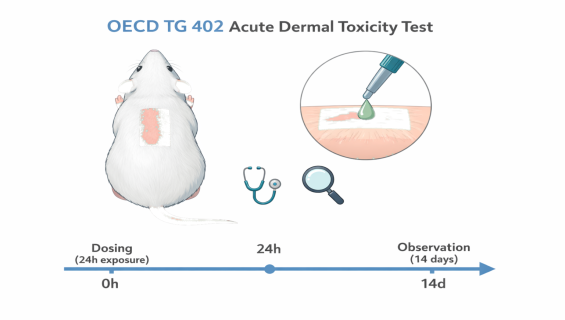
To evaluate the systemic toxic effects resulting from a single, short-term (24-hour) dermal application of a test substance. The results are used to determine the toxicity category and estimate the Median Lethal Dose.
Service Highlights & Technical Specifications
-
Precise Dosing Control: Strict management of the skin contact area (typically at least 10% of the body surface area) and application conditions.
-
Professional Clinical Observation: Intensive 14-day observation by experienced toxicologists to record changes in fur, skin, eyes, respiratory patterns, and behavior.
-
Comprehensive Data Integrity: Detailed tracking of body weight changes, clinical signs, and gross necropsy findings.
-
GHS Alignment: Test results directly support classification under the Globally Harmonized System of Classification and Labelling of Chemicals (GHS).
Study Procedure
-
Initial Test: Selection of a starting dose based on available information or range-finding data.
-
Dermal Exposure: The test substance is applied to the prepared skin area for a period of 24 hours.
-
Observation Period: After removal of the substance, clinical observations are continued for at least 14 days.
-
Reporting: Consolidation of all observed data into a formal GLP-compliant report, including $LD_{50}$ estimations and clinical summaries.
Targeted Industries
-
Safety Data Sheet (SDS/MSDS) Preparation
-
Pesticide and Environmental Agent Registration
-
Industrial Raw Materials and Specialty Chemicals Classification
-
Global Regulatory Submissions (e.g., REACH, TSCA)
- Test species
Narl:LE Rat
-
The objective of this study is evaluation of dermal toxicity caused by test substance in rats. The rats will be exposed to the test substance by dermal application and observed for at most 14 days. The mortality and the changes of apperances will be recorded. According to the results of this test, the category of Globally Harmonized System of Classification and Labelling of Chemicals (GHS) will be defined.
Click to Pay
