Predicting Immune Risks: An In-depth Analysis of OECD TG 404 Skin Sensitization
On the path to product R&D and regulatory compliance (such as Taiwan TFDA, EU MDR, or US FDA), safety assessment is an indispensable milestone. For medical devices, chemicals, or cosmetic raw materials that come into contact with human skin, evaluating "Skin Irritation" and "Skin Corrosion" is typically the primary step.
What is OECD TG 404?
OECD TG 404 is a Test Guideline published by the Organization for Economic Co-operation and Development (OECD). It is primarily used to evaluate the acute effects of a substance on the skin. The assessment is divided into two major categories:
-
Dermal Corrosion: Refers to the production of irreversible damage to the skin, such as necrosis through the epidermis and into the dermis, following the application of a test substance.
-
Dermal Irritation: Refers to the production of reversible inflammatory changes in the skin, such as erythema (redness), eschar, or edema (swelling).
Why is this Test Vital for Your Product?
For medical device manufacturers and chemical producers, data from TG 404 is a core component of the Risk Management Report (ISO 14971) and Biocompatibility Evaluation (ISO 10993):
-
Regulatory Necessity: It is a fundamental technical document required when applying for product registration to prove human safety.
-
Labeling Requirements: Experimental results determine whether a product requires warning labels, such as "Irritant."
-
R&D Optimization: Identifying potential material risks early in the development phase prevents high-cost failures during later clinical trial stages.
Test Procedure and Key Highlights of OECD TG 404
To balance scientific rigor with animal welfare (the 3Rs principles), TG 404 adopts a Tiered Testing Strategy:
1. Initial Assessment (Pre-test)
Before conducting animal studies, we analyze the physical and chemical properties of the substance (e.g., whether the pH is extreme, such as $\le 2.0$ or $\ge 11.5$). If a substance has already been shown to be corrosive through in-vitro tests, no further animal testing is required.
2. In-vivo Study
-
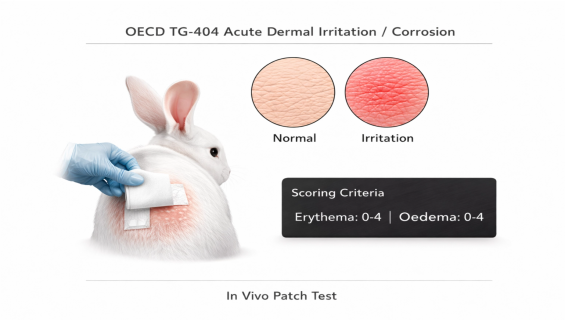
Test Subjects: Healthy adult albino rabbits (typically New Zealand Whites) are used.
-
Application: The test substance is applied to a small area of the rabbit's shorn skin and covered with a gauze patch.
-
Observation Period: Responses are graded at 1, 24, 48, and 72 hours after patch removal. If delayed responses occur, the observation period may be extended up to 14 days to determine the reversibility of the lesions.
3. Scoring Criteria (Draize Scoring)
We grade Erythema and Edema on a scale of 0 to 4. The "Primary Irritation Index (PII)" is then calculated to classify the results as:
-
Non-irritant
-
Slightly irritant
-
Moderate irritant
-
Severe irritant
Click to Pay
