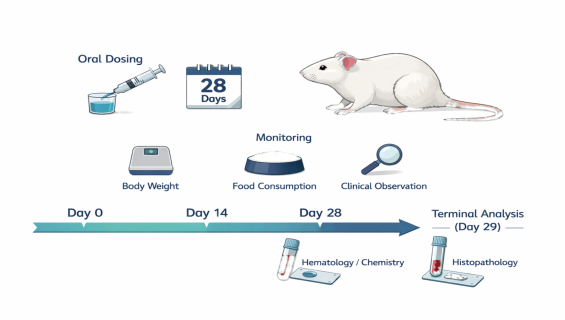
OECD TG 407: Repeated Dose 28-Day Oral Toxicity Study in Rodents
During the intermediate stages of chemical or drug development, understanding the cumulative effects of long-term ingestion on physiological functions is essential. Following OECD TG 407 guidelines and GLP principles, we provide critical sub-acute/sub-chronic toxicity data through a 28-day repeated dosing study to help determine safe exposure levels.
Study Objective
To evaluate the toxicological profile of a test substance administered orally to rodents (typically rats) daily for 28 days. This study identifies target organs, characterizes dose-response relationships, and estimates the No-Observed-Adverse-Effect Level .
Service Highlights & Technical Specifications
-
Versatile Administration Routes: Options include dietary incorporation, drinking water, or oral gavage to suit your substance properties.
-
Comprehensive Physiological Assessment:
-
Clinical Observations: Daily monitoring of behavior, skin, fur, and neurological signs.
-
Hematology & Clinical Chemistry: Assessing liver/kidney functions, metabolic markers, and hematopoietic effects.
-
Functional Observational Battery (FOB): Including sensory reactivity, grip strength, and motor coordination.
-
-
Pathological Identification: Complete gross necropsy, organ weight measurements, and histopathological examinations of tissues.
-
Recovery Period Option: Including recovery groups to assess the reversibility of toxic effects after dosing cessation.
Study Procedure
-
Dose Selection: Utilizing at least three dose levels (High, Mid, Low) and a control group.
-
Continuous Exposure: Daily administration of the test substance for a 28-day duration.
-
Periodic Monitoring: Weekly recording of body weight, food consumption, and clinical symptom scanning.
-
Final Evaluation: Blood/urine sampling followed by comprehensive histopathological analysis.
-
Reporting: Delivering formal GLP-compliant reports suitable for international regulatory submissions.
Targeted Industries
-
New Chemical Notification and Registration (REACH / TSCA)
-
Food Additives and Nutritional Ingredient Safety Assessment
-
Pre-clinical Pharmaceutical Safety Testing
-
Sub-chronic Toxicity Characterization for Pesticides
Click to Pay
