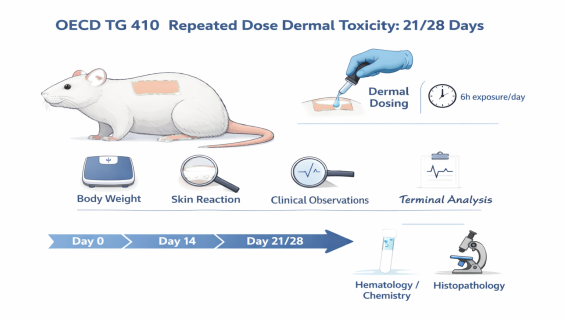
OECD TG 410: Repeated Dose Dermal Toxicity: 21/28-Day Study
For industrial chemicals, pesticides, or personal care ingredients involving frequent skin contact, acute data is insufficient to guarantee safety. Our laboratory adheres to OECD TG 410 guidelines and GLP standards, providing precise repeated dose dermal toxicity evaluations to identify potential health risks from prolonged exposure.
Study Objective
To evaluate the cumulative effects of a test substance administered via the dermal route (daily exposure for at least 6 hours over 21 or 28 days). This study aims to identify target organ toxicity, characterize local skin reactions, and estimate the No-Observed-Adverse-Effect Level (NOAEL).
Service Highlights & Technical Specifications
-
Standardized Dermal Application: Precise control of the application area (approx. 10% of body surface area) using appropriate dressing techniques (occlusive, semi-occlusive, or non-occlusive) to simulate real-world scenarios.
-
Local & Systemic Assessment:
-
Local Assessment: Recording erythema, edema, or other irritant responses at the site of application.
-
Systemic Monitoring: Tracking body weight, food consumption, and clinical signs.
-
-
Clinical Pathology: Comprehensive hematology and clinical biochemistry at the end of the study to assess organ system integrity.
-
Histopathological Analysis: Gross necropsy and histopathology of the treated skin site and major internal organs to detect tissue-level abnormalities.
Study Procedure
-
Experimental Design: Inclusion of at least three dose levels and a control group, with an optional recovery group to assess the reversibility of effects.
-
Dosing Regimen: Daily application to the prepared skin site throughout the 21/28-day period.
-
Observation & Monitoring: Daily clinical observations and weekly measurements of body weight and food intake.
-
Reporting: Consolidation of clinical data, blood chemistry, and pathology into a formal GLP-compliant report.
Targeted Industries
-
GHS Classification and Labeling of Industrial Chemicals
-
Exposure Assessment for Pesticides and Agrochemicals
-
Safety Verification for Long-term Contact Cosmetic Ingredients
-
REACH and International Regulatory Submissions
Click to Pay
