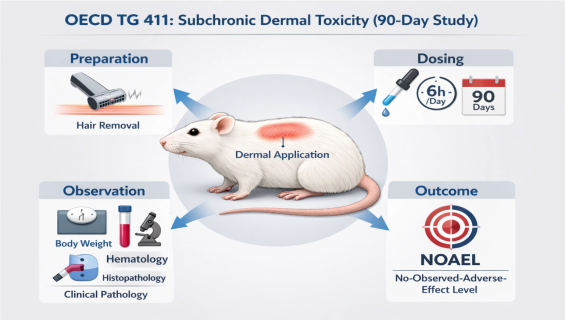
OECD TG 411: Subchronic Dermal Toxicity: 90-Day Study
Short-term studies may not fully reveal potential health risks for products involving chronic or repeated skin contact. Following OECD TG 411 guidelines and GLP principles, we offer a 90-day subchronic dermal toxicity assessment to thoroughly investigate cumulative physiological effects, ensuring your product meets the highest international regulatory standards.
Study Objective
To characterize the toxicological profile of a test substance administered via the dermal route (typically 6 hours per day, at least 5 days a week, for 90 days). This study identifies target organs, observes potential cumulative toxicity, and provides essential data for establishing long-term safety limits, such as the No-Observed-Adverse-Effect Level .
Service Highlights & Technical Specifications
-
Chronic Exposure Simulation: Rigorous dosing protocols that precisely control skin contact area and application pressure to simulate real-world long-term exposure scenarios.
-
In-depth Clinical Analysis:
-
Clinical Pathology: Detailed hematology and biochemistry at various intervals to assess the integrity of hepatic, renal, hematopoietic, and endocrine systems.
-
Urinalysis: Monitoring the impact of substance metabolism on the urinary system.
-
-
Expert Histopathology: Comprehensive gross necropsy and microscopic tissue examination of major internal organs and treated skin sites by senior pathologists.
-
Functional Assessment: Recording sensory reactivity, motor activity, and behavioral changes to detect potential signs of neurotoxicity.
Study Procedure
-
Dose Level Selection: Establishing appropriate dose levels for the 90-day study based on results from 28-day preliminary tests.
-
Continuous Dosing Phase: Daily dermal applications maintained over a 90-day period within a GLP-controlled environment.
-
Dynamic Monitoring: Continuous tracking of body weight fluctuations, food consumption efficiency, and clinical signs.
-
Comprehensive Reporting: Integrating all physiological, chemical, and pathological data into a formal toxicological report compliant with international regulatory standards.
Targeted Industries
-
Safety Assessment of High Production Volume (HPV) Chemicals
-
Registration of Long-term Contact Pesticides and Agrochemicals
-
Environmental Health Risk Identification for Specialized Industrial Materials
-
International Regulatory Submissions (e.g., EU REACH, US TSCA)
Click to Pay
