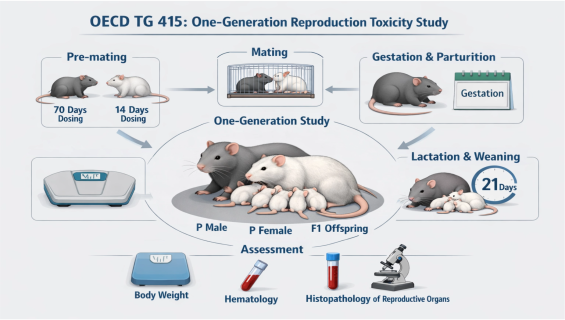
OECD TG 415: One-Generation Reproduction Toxicity Study
Reproductive toxicity assessment is a vital component of ensuring the long-term safety of chemical substances. Adhering to OECD TG 415 guidelines and GLP principles, we provide comprehensive one-generation reproduction toxicity testing services to systematically analyze the effects of test substances on male and female reproductive functions, embryonic development, and early offspring growth.
Study Objective
To evaluate the overall effects of a test substance on the reproductive system, including gonadal function, estrous cycles, mating behavior, conception, parturition, maternal lactation behavior, and the subsequent growth and development of the offspring.
Service Highlights & Technical Specifications
-
Full Life-Cycle Coverage: The dosing period covers the parental (P) generation through pre-mating (10 weeks for males, 2 weeks for females), gestation, and lactation, continuing until the offspring (F1) are weaned.
-
Reproductive Performance Characterization: Precise recording of mating and fertility indices, and gestation length to assess interference with fertility.
-
Offspring Developmental Monitoring: Tracking survival rates, body weight fluctuations, and sex ratios, alongside observations for external malformations or developmental delays.
-
Pathology & Histology Evaluation: Detailed gross necropsy, organ weight measurement, and histopathological examination of parental reproductive organs (e.g., testes, ovaries, epididymides).
Study Procedure
-
Parental Dosing (P Generation): Administration of the test substance via diet, drinking water, or gavage to ensure steady-state exposure.
-
Mating & Gestation: 1:1 pairing, with continuous dosing confirmed from conception through parturition.
-
Lactation & Offspring (F1 Generation): Recording live birth counts and weights, and monitoring offspring health throughout the nursing period.
-
Terminal Evaluation: Following weaning, parental animals and selected offspring undergo necropsy and comprehensive pathological analysis.
-
Reporting: Delivery of formal GLP-compliant reports featuring robust statistical data and toxicological interpretations for international regulatory submissions.
Targeted Industries
-
Chemical Safety Assessment (REACH)
-
Reproductive Impact Identification for Pesticides and Agrochemicals
-
Safety Testing for Food Contact Materials and Additives
-
Pre-clinical Pharmaceutical Safety Research
Click to Pay
