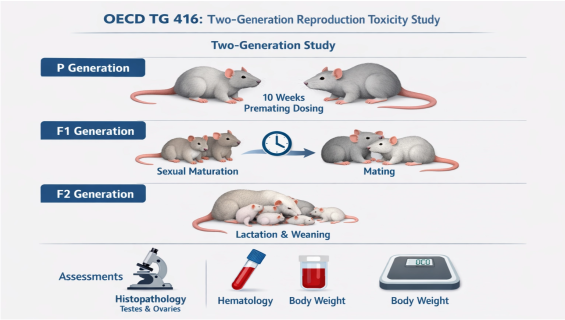
OECD TG 416: Two-Generation Reproduction Toxicity Study
When chemical substances may pose intergenerational risks, an in-depth two-generation study is the gold standard for risk assessment. Adhering to OECD TG 416 guidelines and GLP principles, we provide cross-generational observations to precisely evaluate the effects of test substances on reproductive function and development across parental, first, and second generations.
Study Objective
To systematically evaluate the effects of a test substance on male and female reproductive systems, including gonadal function, estrous cycles, mating behavior, conception, parturition, lactation, and the growth of offspring. A key focus is the reproductive performance of the first-generation (F1) offspring as they reach adulthood, identifying potential intergenerational toxicities.
Service Highlights & Technical Specifications
-
Multi-Generational Tracking: Dosing begins with the parental (P) generation and continues through the full development and reproductive cycle of the F1 generation, concluding at the weaning of the second-generation (F2) pups.
-
Comprehensive Reproductive Indices: Detailed recording of mating and fertility indices, live birth counts, sex ratios, and survival rates across all generations.
-
Sperm and Estrous Cycle Evaluation: Includes sperm count, morphology, and motility analysis, alongside histological monitoring of estrous cycles to provide mechanistic data.
-
Advanced Histopathology: Detailed gross necropsy and histopathological examination of reproductive organs (testes, epididymides, prostate, ovaries, uterus, etc.) for animals in all generations.
Study Procedure
-
Parental (P) Exposure: Continuous administration prior to mating (10 weeks for males, 2 weeks for females), followed by mating, gestation, and lactation.
-
First Generation (F1) Maturation: Selection of F1 individuals post-weaning for continued dosing until sexual maturity and subsequent mating.
-
Second Generation (F2) Observation: Evaluating the development and clinical signs of F2 offspring produced by the F1 generation until weaning.
-
Data Integration: Comparing toxicological findings across generations to determine if the substance possesses cumulative or transgenerational reproductive risks.
Targeted Industries
-
High Production Volume (HPV) Chemical Safety Assessment (REACH Annex X)
-
Comprehensive Reproductive Risk Identification for Pesticides
-
Long-term Safety Verification for Food Additives and Specialized Ingredients
-
Pre-clinical Pharmaceutical Safety Assessment
Click to Pay
