OECD TG 423: Acute Oral Toxicity - Acute Toxic Class Method
Precise acute toxicity classification is a fundamental requirement for safety and regulatory compliance before a product enters the market. Adhering to OECD TG 423 guidelines and GLP principles, we offer professional "Acute Toxic Class Method" testing. Through its stepwise dose design, this method accurately identifies GHS toxicity categories while minimizing animal usage, making it the ideal choice for your regulatory submissions.
Study Objective
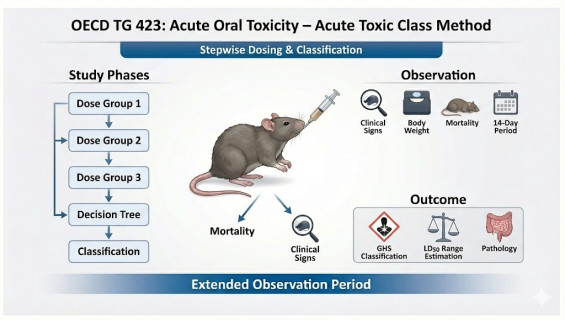
Utilizing a stepwise dosing procedure to observe health effects and mortality following a single oral administration of a test substance. This study determines the acute toxicity class (Category 1 to Category 5) and estimates the cut-off values according to GHS standards.
Service Highlights & Technical Specifications
-
Stepwise Testing Procedure: Depending on the expected toxicity, a starting dose (5, 50, 300, or 2000 mg/kg) is selected. Successive steps are determined by the outcomes of previous stages, ensuring both accuracy and efficiency.
-
Global Credibility: Test results align directly with UN GHS classification criteria and are widely accepted by REACH, EPA, and chemical management authorities worldwide.
-
Rigorous Clinical Observation: During the 14-day observation period, detailed records are maintained regarding changes in skin, fur, eyes, respiration, circulation, and autonomic nervous system activity.
-
Comprehensive Pathological Analysis: Gross necropsy is performed on all test animals to analyze internal organ damage, providing in-depth toxicological insights.
Study Procedure
-
Selection of Starting Dose: Determining the most appropriate starting dose based on available information or range-finding data.
-
Sequential Dosing: Each step utilizes three animals of the same sex (typically females) for dosing and observation.
-
Decision-Tree Evaluation: Based on the mortality rate at each dose level, the study proceeds to either finish, repeat the dose, or move to the next dose level.
-
Comprehensive Reporting: Consolidation of all observational data and necropsy findings into a formal GLP report, including GHS classification recommendations.
Targeted Industries
-
Toxicity Classification for Safety Data Sheets (SDS/MSDS)
-
Registration of Pesticides, Environmental Agents, and Specialty Chemicals
-
Global Regulatory Registration of Industrial Raw Materials and Intermediates (e.g., REACH, TSCA, K-REACH)
-
Initial Safety Screening for Newly Developed Chemical Ingredients
- Test species
Narl:LE Rat
- Introduction
The objective of this study is evaluation of the acute oral toxicity caused by test substance in rats. The test substance will be administered to rats by oral route. The mortality and the changes of apperances will be recorded for 14 days. According to the results of this test, both median lethal oral dose(LD50) and the category of Globally Harmonized System of Classification and Labelling of Chemicals (GHS) will be defined.
Click to Pay
