OECD TG 425: Acute Oral Toxicity: Up-and-Down Procedure (UDP)
Achieving a balance between high scientific precision and the 3Rs of animal welfare, OECD TG 425 stands as the gold standard for acute toxicity assessment. Adhering to GLP principles, we utilize the "Up-and-Down Procedure" to provide the most accurate $LD_{50}$ estimations and GHS classifications while significantly minimizing animal usage.
Study Objective
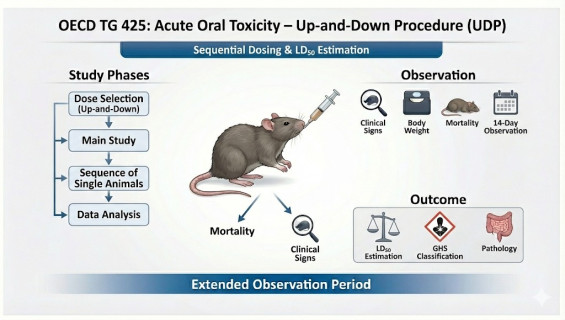
To precisely determine the Median Lethal Dose and its confidence intervals through a sequential single-animal dosing procedure. This study provides more detailed dose-response data compared to other acute toxicity methods.
Service Highlights & Technical Specifications
-
Superior Animal Efficiency: By dosing only one animal at a time and adjusting the subsequent dose based on the outcome, this method requires far fewer animals than traditional tests.
-
Precise Estimation: Utilizing specialized statistical software (AOT programs) to calculate values with scientific confidence intervals, offering more granularity than class-based methods.
-
Broad Regulatory Acceptance: Highly favored by the US EPA, FDA, and REACH (EU), making it ideal for substances requiring high-precision data.
-
Comprehensive Clinical Monitoring: Includes a 14-day observation period and final gross necropsy to document the effects of the test substance on internal organs.
Study Procedure
-
Initial Dose Selection: Setting the starting dose based on an estimate of the from available data.
-
Sequential Testing: If the animal survives, the next animal receives a higher dose; if it dies, the dose is decreased for the next animal.
-
Stopping Criteria: The study concludes automatically once statistical power requirements or sample limits are met.
-
Statistical Reporting: Final results are processed using the Maximum Likelihood Method to generate a formal GLP-compliant report.
Targeted Industries
-
Chemical Registration requiring precise values
-
Safety Identification for Pesticides, Pharmaceuticals, and Specialty Chemicals
-
Precision Classification for High-Toxicity Substances
-
Global Regulatory Submissions (US EPA, REACH, OECD System)
Click to Pay
