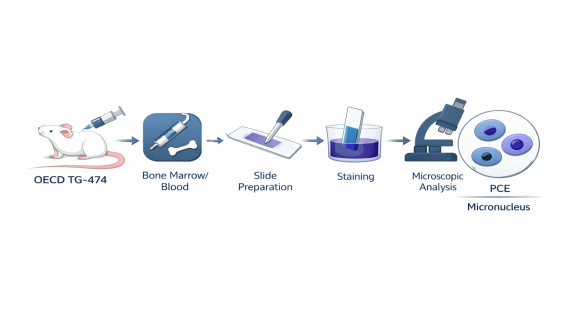
OECD TG-474 — Mammalian Erythrocyte Micronucleus Test
When evaluating the genetic safety of a substance, in vitro tests alone are often insufficient. OECD TG-474 (Micronucleus Test) is the internationally recognized gold standard for assessing whether a chemical induces chromosomal damage "in vivo." It accurately captures the mutagenic risk posed to bone marrow cells after a substance enters systemic circulation, serving as a pivotal benchmark for genetic safety.
1. Why Does Your Product Require an OECD TG-474 Study?
As a critical in vivo validation step in the genotoxicity "Battery Test," the Micronucleus Test offers decisive value:
-
Overcoming in vitro Limitations: In vitro assays cannot fully replicate complex physiological metabolism and distribution. TG-474 reflects the Absorption, Distribution, Metabolism, and Excretion (ADME) of a substance, providing biologically relevant data.
-
Detecting Multiple Genetic Lesions: This test simultaneously detects clastogens (causing chromosome breakage) and aneugens (causing chromosome loss). By observing micronucleus formation in immature erythrocytes, it identifies interferences in cell division.
-
Golden Evidence for Regulatory Submission: For medical devices (ISO 10993-3), New Drug Applications (IND/NDA), or high-tonnage chemicals (REACH), the micronucleus test is a mandatory genotoxicity data requirement for global regulatory agencies.
2. Technical Depth: Principles of Micronucleus Formation
The scientific rigor of OECD TG-474 lies in its dynamic monitoring of the erythropoiesis process:
-
Origin of the Micronucleus: When bone marrow cells are damaged, chromosomal fragments or whole chromosomes lag behind during cell division and remain outside the daughter nuclei, forming a small "micronucleus."
-
Observation Target — Immature Erythrocytes (PCEs): Since erythrocytes expel their main nucleus during maturation, the presence of a micronucleus in a "Polychromatic Erythrocyte (PCE)" serves as definitive evidence of genetic damage sustained during the bone marrow development stage.
-
Assessment of Cytotoxicity: By calculating the ratio of PCEs to total erythrocytes (NCEs), we evaluate the degree of bone marrow suppression caused by the test substance, ensuring the sensitivity of the assay.
3. Your Premier Regulatory and Testing Partner
Executing OECD TG-474 requires high-precision microscopy or automated flow cytometry analysis. We provide:
-
GLP-Compliant Excellence: Our protocols strictly adhere to OECD and ICH guidelines. Reports carry international mutual recognition (MAD), facilitating seamless acceptance by global regulators.
-
Versatile Dosing and Sampling Strategies: Tailored to the properties of your substance, we offer single or multiple dosing regimens, with precise sampling at the critical 24- and 48-hour windows.
-
Expert Data Interpretation: Our toxicology experts assist in analyzing the statistical significance of micronucleus frequency and provide strategic advice for responding to positive results, ensuring your regulatory pathway remains on track.
Click to Pay
