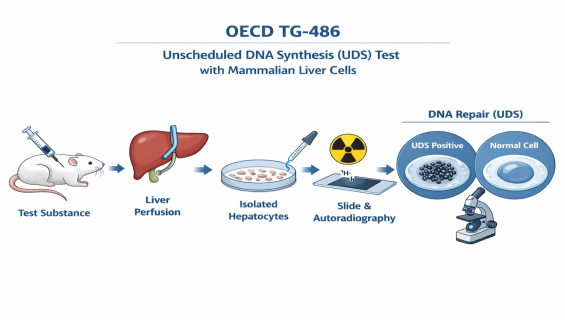
OECD TG-486 — Unscheduled DNA Synthesis (UDS) Test with Mammalian Liver Cells in vivo
In the complex hierarchy of toxicological evaluation, when in vitro results are ambiguous or when organ-specific validation is required, OECD TG-486 (UDS Test) stands as the authoritative tool for identifying DNA damage and repair in the liver. It precisely detects whether a chemical induces "unscheduled" DNA repair activity after entering the primary metabolic hub—the liver.
1. Why Does Your Product Require an OECD TG-486 Study?
The UDS assay typically functions as a secondary validation tool, playing a decisive role in the following regulatory pathways:
-
Clarifying Hepatocarcinogenic Risk: If sub-chronic studies indicate liver damage or if in vitro mutagenicity (Ames) tests are positive, TG-486 directly determines if DNA damage occurs within the in vivo hepatic environment.
-
Evaluating Post-Metabolic Genotoxicity: The liver is the body's most critical metabolic organ. Many chemicals only become toxic after enzymatic activation. The UDS assay accurately captures the impact of these "metabolically activated" toxins on the genome.
-
Complementing the Genotoxicity Battery: For pharmaceutical R&D, specialized pesticides, and industrial chemicals, UDS serves as a strategic tool to strengthen regulatory evidence and resolve technical Additional Information (AI) requests when conventional micronucleus tests are insufficient.
2. Technical Depth: Principles of DNA Repair and Autoradiography
The core value of OECD TG-486 lies in its ability to detect "unexpected" DNA synthesis:
-
Defining UDS: DNA synthesis occurring outside the normal cell division cycle (S-phase) indicates that the cell is actively repairing damaged DNA segments.
-
Radionuclide Labeling: Following administration of the test substance, hepatocytes are isolated and exposed to radiolabeled thymidine ($^3H-TdR$). If DNA damage has occurred, the cells incorporate these radioactive labels during the repair process.
-
Autoradiography Technique: Using microscopy, the density of silver grains within the hepatocyte nucleus is observed. The Net Grain Count (nuclear grains minus cytoplasmic background) is the key indicator for judging the severity of DNA damage.
3. Your Premier Regulatory and Testing Partner
Executing OECD TG-486 requires sophisticated hepatocyte isolation techniques and precise radioactive detection expertise. We provide:
-
GLP-Compliant Excellence: Our protocols strictly adhere to OECD guidelines, with data carrying international mutual recognition (MAD), ensuring seamless acceptance by global regulators.
-
Precise Sampling Window Control: Since UDS is a transient phenomenon, we meticulously manage the dual sampling windows at 2-4 hours and 12-16 hours post-administration to ensure no damage signals are missed.
-
In-depth Toxicological Integration: Our expert team assists in cross-referencing UDS data with other genotoxicity results, providing a comprehensive and persuasive environmental and health risk assessment report.
Click to Pay
