OECD TG 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test
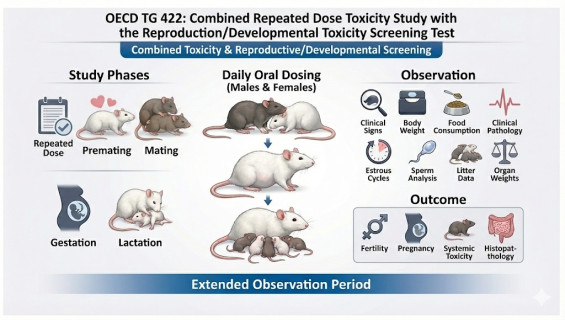
For those seeking a balance between regulatory compliance and time efficiency, OECD TG 422 offers a highly advantageous solution. Adhering to GLP principles, we combine "Repeated Dose Toxicity" and "Reproduction/Developmental Screening" into a single comprehensive study, minimizing animal usage while providing a multidimensional toxicological profile for your product.
Study Objective
This study is designed to simultaneously evaluate the systemic toxicity resulting from repeated exposure and potential risks to reproductive performance (male and female fertility) and early offspring development.
Service Highlights & Technical Specifications
-
High Information Integration: A single study provides data on repeated dose toxicity, neurotoxicity screening, reproductive toxicity, and developmental effects.
-
Systemic Toxicity Evaluation: Includes detailed clinical observations, body weight/food consumption tracking, hematology, and clinical biochemistry.
-
Reproductive & Developmental Screening: Monitoring parental mating performance, conception rates, parturition, and the survival and growth of offspring post-birth.
-
Functional Observational Battery (FOB): Incorporating sensory reactivity and motor activity tests to assess potential neurotoxic effects.
-
Cost-Effective Compliance: Optimizes resources and serves as a core requirement for REACH Annex VIII (10-100 tonnes/year).
Study Procedure
-
Parental Exposure: Males are dosed for at least 28 days; females are dosed starting 2 weeks prior to mating and continuing through gestation up to post-natal day 13.
-
Clinical & Functional Examination: Clinical symptom scanning and Functional Observational Battery (FOB) tests are conducted during the dosing period.
-
Reproductive Phase: Recording indices for mating, conception, parturition, and lactation.
-
Terminal Evaluation: Blood and urine analysis, followed by comprehensive gross necropsy and histopathological examination of parental organs and offspring.
Targeted Industries
-
Regulatory Chemical Notification (REACH Annex VIII)
-
Integrated Risk Assessment in Early Product Development
-
Preliminary Toxicity Characterization for Pesticides and Industrial Chemicals
-
R&D Projects requiring rapid acquisition of multi-dimensional toxicological data
Click to Pay
